New UNC Charlotte study using ultra-sensitive microscope reveals how just three molecules can organize the epigenome

In the microscopic world inside a stem cell, some of the most important decisions about life’s earliest stages come down to only a handful of molecules. A new study from UNC Charlotte shows that just three molecules of a rare protein can spark the formation of massive gene‑silencing structures, helping cells choose what they will ultimately become.
The work, published in Molecular Cell, comes from the lab of Xiaojun Ren, associate professor and Irwin Belk Distinguished Scholar of Biology. Ren has spent years trying to understand how Polycomb proteins, which are key regulators of cell identity, organize the genome. The problem: some of these proteins are so scarce that traditional tools simply cannot detect them.
“This isn’t like finding a needle in a haystack. This is like finding a needle in the ocean.”
–Xiaojun Ren, associate professor and Irwin Belk Distinguished Scholar of Biology
A rare protein with an outsized influence
Stem cells can turn into anything: a brain cell, a muscle fiber, a blood cell, or anything the protein tells them to be. Polycomb complexes are essential for keeping certain genes switched off so cells can specialize. But scientists have long puzzled over how these complexes assemble into the large, droplet‑like “condensates” that silence genes across the genome.

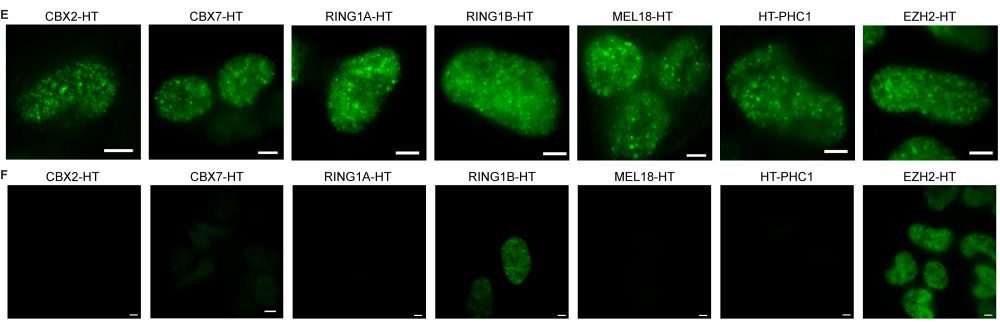
Ren’s team cracked the mystery using an ultra‑sensitive live-cell single‑molecule microscope, technology that is not available at many labs worldwide. The instrument allows researchers to count individual proteins inside living cells, one molecule at a time.
With this imaging technology, the team made a surprising discovery: In mouse embryonic stem cells, Polycomb condensates form around a tiny core of just three molecules of a protein called CBX2.
“It’s remarkable,” Ren said. “CBX2 is one of the least abundant Polycomb proteins in stem cells, yet it can dictate the assembly of these large regulatory structures and act as a seed for the entire condensate. CBX2 is doing far more with far less than anyone expected.”
Those three molecules act like the first bubbles in a pot of boiling water — small, fleeting, but powerful enough to trigger a cascade. As more components join in, the bubbles merge into a single, larger droplet. In the same way, CBX2’s tiny clusters recruit additional Polycomb complexes, gradually building the gene‑repressive hubs that shape the epigenome.
Why this matters

Polycomb dysfunction is linked to developmental disorders and cancers. By revealing how condensates form — and just how fragile that process can be — the study offers a new lens for understanding how gene‑silencing programs are built, maintained and sometimes derailed.
It also underscores the power of cutting‑edge imaging technologies. Without UNC Charlotte’s ultra‑sensitive single‑molecule microscope, these three‑molecule “seeds” would have remained invisible.
Ren’s lab plans to continue exploring how condensates change as cells mature and how disruptions to these early molecular events might contribute to disease.
In the vast ocean of the cell, finding three molecules may seem impossible. The microscope, in the capable hands of the Ren lab and their research team, uncovered how molecules can reveal how life organizes itself from the ground up.
How three molecules reshape the genome
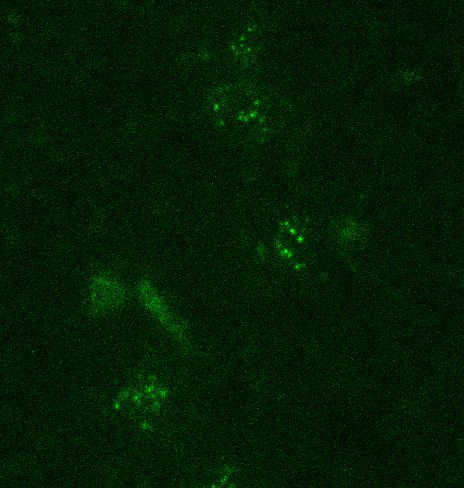
Once Ren’s team realized that tiny CBX2 clusters could act as “starter bubbles” for gene‑silencing droplets, they dug deeper into what those bubbles actually do. They found that these little clusters act like magnets, attracting other components. Even though CBX2 is one of the rarest proteins in the cell, its small groups are powerful enough to pull in other major players in the Polycomb system: proteins that help shut down genes so a cell can stay on the right developmental path.
Together, these proteins form busy control hubs that help determine which genes remain silent. And CBX2, despite being scarce, plays a surprisingly central role in steering where these silencing marks land across the genome.
To understand how CBX2 guides this process, the team looked across the entire genome. They found that CBX2 tends to settle at the very spots where gene‑silencing marks first begin to form. When the researchers removed CBX2, those marks and the proteins that place them wandered to the wrong locations. The result was a weakening of gene repression at some of the most important developmental genes.
As stem cells begin to specialize, the droplets grow and change with the cell’s identity. For example, early neural cells contain about 15 CBX2 molecules in each droplet, which is five times more than in embryonic stem cells. This is a sign that as cells choose their future, they also rebuild the machinery that keeps certain genes turned off.
In other words, without CBX2 acting as the tiny spark that starts the whole process, the cell’s gene‑silencing system loses its map.
A mutant that reveals CBX2’s true power
To understand just how important CBX2’s “clustering” ability is, Ren’s team created a special version of the protein that was missing this one feature. Think of it as CBX2 with its hands tied: it could still latch onto DNA, but it was unable to link arms with other CBX2 molecules the way the normal protein does.
What happened next made the answer clear.
Inside living cells, the altered CBX2 could not form the tiny starter clusters that kick off condensate formation. In lab tests, it required more than 100 times the usual amount of protein before it even began to clump. And without those early clusters, the cell’s gene‑silencing marks drifted to the wrong places, piling up in dense stretches of DNA instead of the regions they are meant to regulate.
The ripple effects were striking. When the team encouraged the cells to start becoming specialized cell types, the ones carrying the altered CBX2 struggled. Their early embryonic structures barely grew. They had trouble producing neural precursor cells. And the number of cells showing key markers of neural identity dropped sharply.
“Without CBX2’s ability to self‑cluster, the cells simply cannot execute their developmental programs,” Ren said. “Just as a raindrop needs a tiny speck of dust to form in a humid sky, these three molecules act as the seeds that pull the cell’s Polycomb proteins together into a functional droplet.”
A new way to think about how these droplets form

For years, scientists assumed that Polycomb condensates formed the same way oil droplets separate from water — a classic process called liquid‑liquid phase separation. But Ren’s team found that this old model does not hold up. With only about three molecules of CBX2 at the center of each condensate in stem cells, there simply are not enough molecules to form the dense networks that traditional phase separation requires.
Instead, the researchers propose a new explanation. In their model, CBX2 acts like the first tiny grain around which everything else gathers. It attaches to specific spots on the genome, creating a small landing pad. That pad then attracts other Polycomb proteins, which help bridge nearby stretches of DNA. As more proteins join in, the structure grows — much like small bubbles merging into a larger droplet.
This new framework helps explain how a protein that’s incredibly rare can still organize huge stretches of the genome. It also brings together years of scattered observations into a single, coherent picture of how gene‑silencing structures take shape inside living cells.
More about the Ren lab
The Ren laboratory relocated to UNC Charlotte in the summer of 2024 and focuses on epigenetic mechanisms, genome organization, single-molecule imaging, liquid-liquid phase separation and Polycomb proteins. Their work is supported in part by grants from NIH R01GM135286, and funds from the University of North Carolina at Charlotte. Ren was previously an associate professor at the University of Colorado Denver, so this research was in part supported by CU Denver prior to this move.

Photos by Kat Lawrence. Molecular images courtesy of the Ren lab.