UNC Charlotte Truman Lab publishes new insights into cellular heat shock response in Nature Communications

A new study from UNC Charlotte’s Truman Lab has been published in Nature Communications, offering a deeper understanding of how cells respond to heat-induced stress.
The paper, “Mechanosensor-mediated Hsp70 phosphorylation orchestrates the landscape of the heat shock response,” examines a novel key regulatory mechanism that rapidly activates the cellular defense to heat shock.
“This study reveals a fast ‘first step’ in the heat shock response. Instead of waiting for widespread protein damage, cells can sense heat-induced membrane stretch and rapidly modify Hsp70, allowing the cell to launch a coordinated protective program within minutes,” said Andrew Truman, Ph.D., the Truman Lab’s principal investigator, professor of biological sciences and associate chair for research in the Klein College of Science.
From Ph.D. Research to Publication
The article’s first author, Siddhi Omkar, Ph.D., began the project in 2020 as part of her doctoral research in the Truman Lab.
Data generated through Omkar’s research contributed significantly to the lab’s 2024 $1.28 million grant from the National Institute of General Medical Sciences (NIGMS), part of the National Institutes of Health (NIH).
Omkar defended her thesis in November 2024, just months after the birth of her son, before returning to the lab as a postdoctoral researcher. The team submitted the manuscript to Nature Communications in January 2025, and after peer review and revisions, it was accepted in November 2025.


A Cellular ‘Emergency Response Center’
The research team discovered that a single phosphorylation event on the Hsp70 protein functions like a 911 call, triggering multiple protective pathways within minutes of heat shock.
“I compare it to a city emergency department,” Omkar explained. “You receive a phone call about an emergency, and suddenly a number of other departments start working together. One single phosphorylation or one single PTM controls a number of different fundamental events inside the cell, all coordinating to help the cell protect itself from the heat.”

Decoding the ‘Chaperone Code’
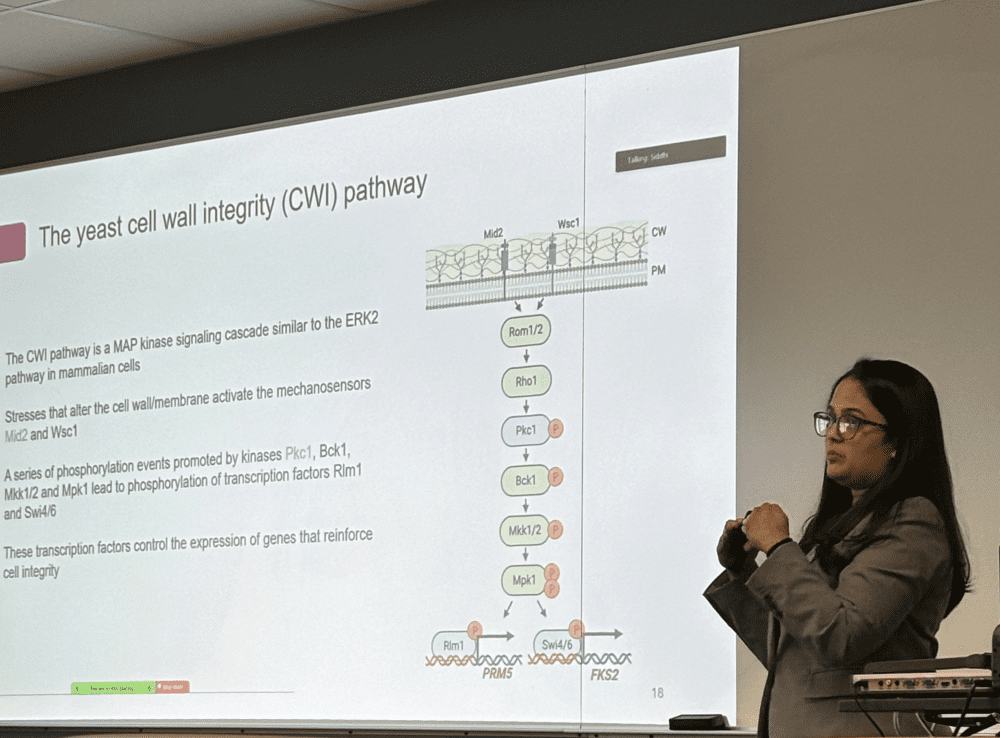
The Truman Lab studies Hsp70, which is found in nearly all forms of life from bacteria to humans. Hsp70 works like a mechanic, making sure proteins fold into the right shapes and do not clump together. It also helps cells deal with proteins that are damaged, either by helping fix them or by directing them to be broken down and recycled.
This is important because Hsp70 can influence disease in more than one way. Many cancer cells depend on Hsp70 to help stressed or altered proteins keep working, which can support tumor growth.
In contrast, in brain diseases like Huntington’s, Alzheimer’s and ALS, Hsp70 can help reduce the buildup of harmful protein clumps that damage cells. Which effect you see depends on the type of cell and which proteins Hsp70 is working on.
A major focus of the lab is understanding how small chemical “tags” (Post-translational modifications or PTMs) added to Hsp70 act like control switches that change how it behaves. The lab calls this collection of tags the Chaperone Code. By learning how these switches tune Hsp70, the Truman Lab aims to reveal new ways to understand disease and potentially guide future treatments.

Understanding Heat Stress Responses
During her Ph.D., Omkar set out to address a fundamental question in biology: How do cells respond so rapidly to heat stress?

Her findings reveal how the Chaperone Code fine‑tunes proteostasis, which is the delicate balance of protein folding, stability and degradation. Together, the results present a unified model of the global heat shock response in yeast, governed by Hsp70‑mediated signaling.
Future Implications
Because protein misfolding underlies many cancers and neurodegenerative diseases, the work may have broad biomedical impacts.
“This particular site is also known to be mutated in cancer, which makes it much more interesting,” Omkar said. “We can use this as a targeted cancer therapy, which is just one very important implication.”
Omkar plans to continue her postdoctoral work in the Truman Lab for a few years before moving into industry.
“Right now I am focusing on other sites and other chaperone codes that are of interest to me,” she said. “Eventually I would like to transition into industry to do something that is related to disease or clinical-level research.”
Collaborative Science
This project was made possible due to extensive internal and external partnerships, with special thanks to the UNC Charlotte Division of Research and the Klein College of Science.
The research team collaborated within the Klein College of Science and beyond. Richard Chi and James (Trey) Grissom in UNC Charlotte’s Chi Lab contributed microscopy expertise, Luca Fornelli and Jake Kline at the University of Oklahoma supported mass spectrometry and proteomics and Diyun Sun and Jared Bard from the Bard Lab at Texas A&M University assisted with protein translation research.
“Looking back from starting this project in 2020 to seeing it published, it’s been an amazing journey with many personal and professional milestones along the way,” said Omkar. “I am incredibly thankful for Dr. Truman’s mentorship, my colleagues in the Truman Lab for their support, and my family’s unwavering encouragement throughout. Our collaborators were instrumental in advancing this research, and I’m excited to continue building on this foundation.”